Ava is seropositive for ACPA and RF – how likely is she to have a poor prognosis?

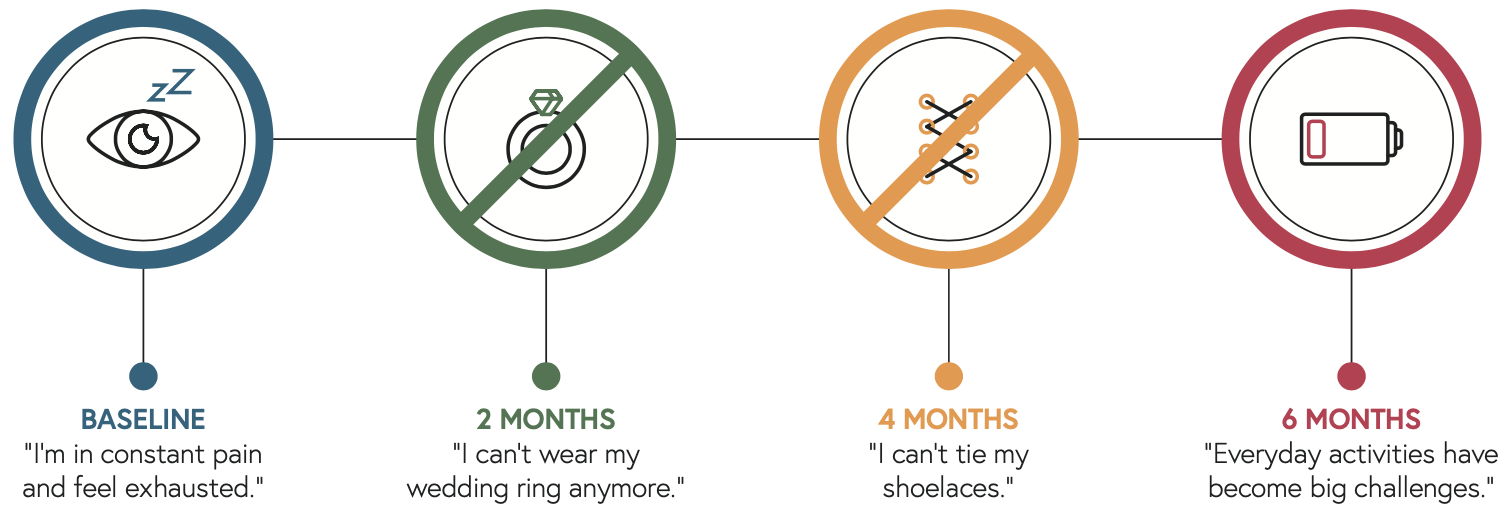
For patients like Ava, time is of the essence1-6
Ava is:
41 year-old POLICE OFFICER

Has a PHYSICALLY demanding job

Has had RA for almost 2 YEARS

Presents with:
BONE EROSIONS

FAMILY HISTORY of aggressive RA
Shared epitope

Did not respond to first biologic


ORENCIA® (abatacept), in combination with methotrexate, is indicated for:7
- the treatment of moderate to severe active rheumatoid arthritis (RA) in adult patients who responded inadequately to previous therapy with one or more
disease-modifying anti-rheumatic drugs (DMARDs) including methotrexate (MTX) or a tumour necrosis factor (TNF)-alpha inhibitor. - the treatment of highly active and progressive disease in adult patients with rheumatoid arthritis not previously treated with methotrexate.
A reduction in the progression of joint damage and improvement of physical function have been demonstrated during combination treatment with abatacept and methotrexate.
How would you rate Ava’s prognosis based on this case information?

What does poor prognosis feel like for an RA patient not responding to treatment?

This patient profile is fictitious, and her clinical characteristics are based on available data in patients with high ACPA serostatus.1–3,5
How likely are you to treat Ava differently from your other RA patients who are not seropositive for ACPA and RF?

Case considerations
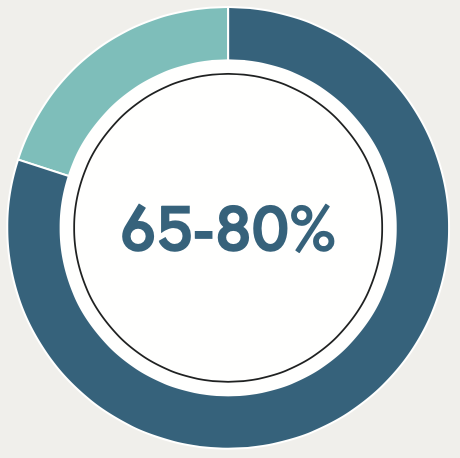
65-80% of RA patients have at least one shared epitope allele 11,13

This patient profile is fictitious, and her clinical characteristics are based on available data in patients with high ACPA serostatus.1–3,5
SE-positive patients are genetically susceptible to form ACPAs and have erosive disease1-6, 8-11
The shared epitope (SE) is a genetic risk factor for highly aggressive erosive RA8-11
- Certain mutations in the human leukocyte antigen (HLA) gene predispose the immune system to more readily bind citrullinated self-proteins and recognise them as foreign8–10
- This results in the formation of pathogenic autoantibodies against those proteins, i.e. ACPAs8–10

At high levels, ACPA titres drive the pathophysiology of RA, which can lead to poor disease prognosis:1–6
- Early and significant bone erosion: Individuals with no clinical signs of RA but found to be seropositive for ACPA presented with lower bone volume, mineral density and eroded surface (p≤0.013 vs seronegative)*1
- Accelerated erosion: ACPA-seropositive RA patients show >2x the level of radiological destruction vs seronegative patients at 2 and 4 years †2

Patients with one or two HLA-DRB1 SE alleles (vs no allele) are nearly 3 to 5 times as likely to be ACPA seropositive and have erosive disease (OR 2.87 [95% CI 1.32, 6.23] for one allele and 5.44 [2.39, 12.39] for two alleles)11
* n=15 ACPA-seropositive vs n=15 ACPA-seronegative subjects.1
† Assessed by the Sharp-van der Heijde method in a single-centred observational study in patients receiving treatment (not controlled for treatment approach).
Out of 454 patients included in this study, 138 had 4-year radiographic data.2
How often do you measure ACPA levels in patients like Ava?

Measure ACPA levels in RA patients like Ava to facilitate and guide appropriate treatment decisions for the long-term5,6
NICE calls for ACPA serostatus testing as soon as possible after diagnosis12

Would you go back and evaluate the serology of Ava at treatment change?

How would you rate Ava’s prognosis based on this case information?
Has any new information changed your original answer?

What steps would you follow next knowing what you know now, for Ava’s management of disease?
Review ACPA levels and status
Full case history of risk factors
Review other diagnostics
Initiate alternative bDMARD

An ACPA lowering treatment may be appropriate5,6
The presence of single or double alleles encoding the shared epitope is associated with ACPA seropositivity and aggressive disease11
ACPA seropositivity drives highly aggressive, erosive RA, and this should be considered when choosing her treatment5,6

ORENCIA®
DAS28-CRP improvement from the first measurement vs baseline, with a comparable onset of action to adalimumab17
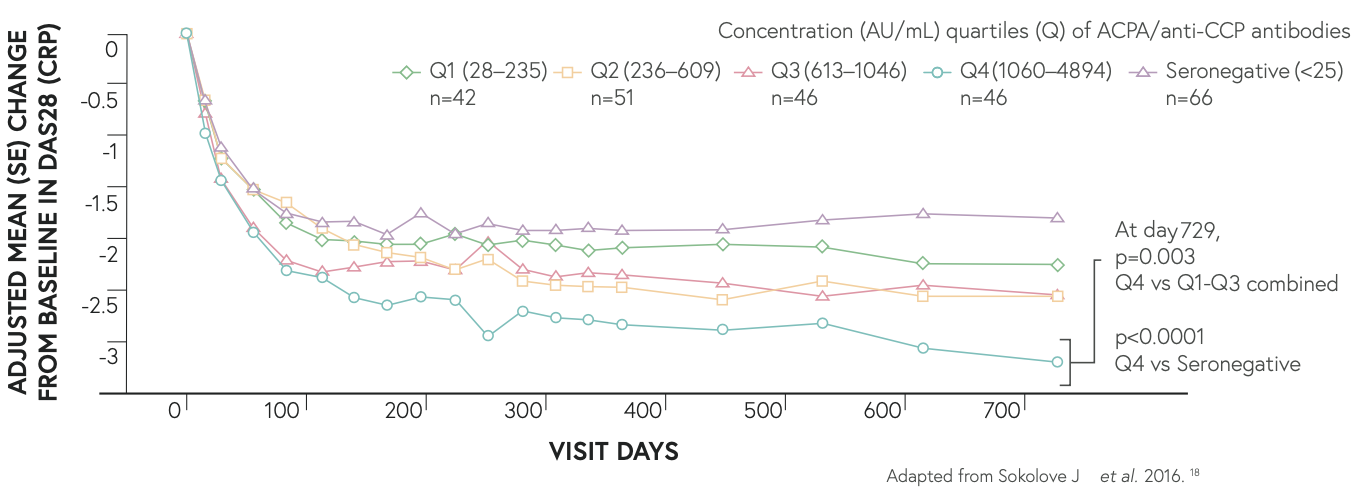
Post-hoc exploratory analyses with ORENCIA® suggest that RA patients with highest ACPA seropositivity have greatest improvements from baseline in DAS28-CRP15
At-risk patients may stay the course with ORENCIA® due to its low rates of drug-related discontinuations7,18

ORENCIA® decreases ACPA levels vs baseline and provides sustained pain relief14-16
ORENCIA® produced a continuous decline in ACPA concentrations over 2 years*14
Post-hoc explanatory analyses suggest that patients with highest ACPA seropositivity have greatest improvements from baseline in DAS28-CRP*15
Pain relief
- ORENCIA® provided sustained improvements in the 100-mm VAS pain score from baseline out to 2 years*16
- A clinically meaningful improvement in patients’ pain was reached after just 15 days of treatment with ORENCIA®
Onset of action
- ORENCIA® reduced DAS28 vs baseline from the first measurement17
- Rate of reduction in DAS28-CRP scores was comparable with ORENCIA® and adalimumab
RA patients with poor prognosis, like Ava, require a treatment that provides balanced efficacy-safety outcomes
*Data from a randomised, Phase 3b, head-to-head study (AMPLE) directly comparing ORENCIA® SC(n=318) with adalimumab SC(n=328), both in combination with MTX, where ~75% of patients in each treatment arm were ACPA seropositive.
The AMPLE study was not designed or powered to draw definitive conclusions on ACPA-based differential treatment effects. Primarily endpoint was treatment non-inferiority by ACR20 response at 1 year.15,17
ORENCIA® may be a suitable choice for patients with poor prognosis, like Ava, due to its acceptable long-term safety profile across clinical studies7,18
Low rates of serious infections and discontinuations due to adverse reactions have been observed in patients treated with ORENCIA®7,18
| Onset of action | Cumulative data | 5-year LTE data (in combination with MTX) |
| Low rates of serious infections | 2.4/100 patient-years (n=7,044 patients; 20,510 patient-years)7 | 2.8/100 patient-years (n=594 patients)*18 |
| Low rates of discontinuations due to adverse reactions | 3% (vs 2% with placebo)7 | 9.8% (n=37/378)*18 |
| Consistent safety profile over 5 years | No increase in the incidence rates of infections, serious infections and autoimmune events in the cumulative study period, and no unexpected events in the LTE, relative to the double-blind period18 | |

RA patients can stay the course with ORENCIA® due to its low rates of discontinuations7,18
Please refer to the Summer of Product Characteristics before prescribing ORENCIA®
*Open-label LTE phase of a placebo-controlled Phase 3 trial (AIM) in which MTX-inadequate responders received ORENCIA® (~10 mg/kg, according to weight range) plus MTX. Data are for all patients who had received at least 1 dose of ORENCIA® in the double-blind or LTE, regardless of treatment group during the double-blind period. 18
Identifying patients with poor prognostic factors such as ACPA seropositivity can help ensure you get it right for RA patients like Ava5,6
Physicians should exercise caution when considering the use of ORENCIA® in patients with a history of recurrent infections or underlying conditions which may predispose them to infections. Patients who develop a new infection while undergoing treatment with ORENCIA® should be monitored closely. Administration of ORENCIA® should be discontinued if a patient develops a serious infection.7

How has this affected your confidence in treating patients with seropositive rheumatoid arthritis?
How likely are you to recommend this case study to a colleague?

References
- Kleyer A et al. Ann Rheum Dis 2014;73(5):854–60.
- van der Helm-van Mil AHM et al. Arthritis Res Ther 2005;7(5):R949–58.
- Sokolove J et al. Arthritis Rheumatol 2014;66(4):813–21.
- Laurent L et al. Ann Rheum Dis 2015;74(7):1425–31.
- Lamerato L et al. J Med Econ 2018;21:231–40.
- van Venrooij WJ et al. Nat Rev Rheumatol 2011;7(7):391–8.
- ORENCIA® Summary of Product Characteristics. Available at www.medicines.org. uk/emc (last accessed: June 2021).
- Busch R, et al. Nat Rev Rheumatol 2019;15:364–81.
- Kampstra ASB et al. Immunogenetics 2017;69:597–603.
- Anderson KM et al. Arthritis Rheum 2016;68(7):1627–36.
- Alemao E et al. Arthritis Rheumatol 2018;70(suppl 10):A71.st improvements fin DAS28-CRP15
- National Institute for Health and Care Excellence. Rheumatoid arthritis in adults: management. NICE guideline. Published: 11 July 2018. Available at www.nice.org.uk/guidance/ng100 (last accessed: June 2021).
- Holoshitz J. Curr Opin Rheumatol 2010;22(3):293–8.
- Connolly S et al. Ann Rheum Dis 2014;73:395.
- Sokolove J et al. Ann Rheum Dis 2016;75(4):709–14.
- Fleischmann R et al. Arthritis Care Res (Hoboken) 2016;68:907–13.
- Schiff M et al. Ann Rheum Dis 2014;73(1):86–94.
- Kremer JM et al. J Rheumatol 2014;41(6):1077–87.
ORENCIA® (abatacept) PRESCRIBING INFORMATION
Consult Summary of Product Characteristics (SmPC) before prescribing
PRESENTATION: Intravenous (I.V.): 250 mg powder for concentrate for solution for I.V. infusion; each mL contains 25 mg of abatacept, after reconstitution.
Pre-filled Syringe: 50 mg, 87.5 mg and 125 mg solution for injection in pre-filled syringe. ClickJect pre-filled pen: 125 mg solution for injection in pre-filled pen
INDICATION:Rheumatoid arthritis (RA) (I.V. infusion, Subcutaneous (SC) pre-filled syringe and pen): Orencia, in combination with methotrexate, is indicated for:
- The treatment of moderate to severe active rheumatoid arthritis in adults who responded inadequately to previous therapy with one or more disease-modifying anti-rheumatic drugs (DMARDs) including methotrexate or a tumour necrosis factor (TNF)-alpha inhibitor.
- The treatment of highly active and progressive disease in adults with rheumatoid arthritis not previously treated with methotrexate.
A reduction in the progression of joint damage and improvement of physical function have been demonstrated during combination treatment with abatacept and methotrexate, see SmPC. Psoriatic Arthritis (PsA)(I.V. infusion, SC pre-filled syringe and pen): Orencia alone or in combination with methotrexate (MTX), is indicated for the treatment of active psoriatic arthritis (PsA) in adults when the response to previous DMARD therapy including MTX has been inadequate, and for whom additional systemic therapy for psoriatic skin lesions is not required. Polyarticular Juvenile Idiopathic Arthritis (pJIA)I.V. infusion: Orencia in combination with methotrexate is indicated for treatment of moderate to severe active pJIA in paediatric patients ≥ 6Y (years of age) who have had an inadequate response to previous DMARD therapy. Orencia can be given as monotherapy in case of intolerance to methotrexate or when treatment with methotrexate is inappropriate. SC pre-filled syringe: Orencia in combination with methotrexate is indicated for the treatment of moderate to severe active pJIA in paediatric patients ≥ 2Y who have had an inadequate response to previous DMARD therapy. Orencia can be given as monotherapy in case of intolerance to methotrexate or when treatment with methotrexate is inappropriate.
DOSAGE AND ADMINISTRATION: Treatment should be initiated and supervised by specialist physicians experienced in the diagnosis and treatment of RA, pJIA or PsA. Orencia 250 mg powder for concentrate for solution for I.V. infusion Adults and elderly dosing (RA or PsA): Patients weighing < 60 kg: 500 mg (2 vials). Patients weighing ≥ 60 kg to ≤ 100 kg: 750 mg (3 vials). Patients weighing > 100 kg: 1000 mg (4 vials). Paediatric dosing (pJIA): Patients ≥ 6 to ≤ 17Y, weighing ≤ 75 kg: 10 mg/kg. Paediatric patients weighing ≥ 75 kg: to be administered adult dosage, not exceeding a maximum dose of 1000 mg. See SmPC for details on 30 minute I.V. infusion. After initial administration, Orencia I.V. should be given at 2 and 4 weeks, then every 4 weeks thereafter. Not recommended in children below ≤ 6Y. Orencia 50 mg, 87.5 mg and 125 mg solution for injection (SC pre-filled syringe) and Orencia 125 mg solution for injection (SC pre-filled pen) Adults and elderly dosing (RA or PsA): Orencia SC may be initiated with or without an I.V. loading dose. Orencia SC should be administered weekly at a dose of 125 mg by SC injection regardless of weight. If a single I.V. infusion is given to initiate treatment (I.V. loading dose before SC administration), the first 125 mg abatacept SC should be administered within a day of the I.V. infusion, followed by the weekly 125 mg abatacept SC injections. Patients transitioning from Orencia I.V. therapy to SC administration should administer the first SC dose instead of the next scheduled I.V. dose. Orencia 50 mg, 87.5 mg and 125 mg solution for injection (SC pre-filled syringe) Paediatric dosing (pJIA): Paediatric patients weighing ≥10 kg < 25 kg: 50 mg. Paediatric patients weighing ≥ 25 kg to < 50 kg: 87.5 mg. Paediatric patients weighing ≥ 50 kg: 125 mg. Patients ≥ 2 to ≤ 17Y with pJIA should be initiated without an I.V. loading dose. The safety and efficacy of Orencia pre-filled pen for SC use in children < 18Y have not been established. Patients switching from Orencia I.V. therapy to SC administration should administer the first SC dose instead of the next scheduled I.V. dose. The continuation of treatment with abatacept should be re-assessed if patients do not respond within 6 months. Special populations: Elderly patients: No dose adjustment is required. Renal and hepatic impairment. No dose recommendations can be made. Paediatric population: The safety and efficacy of IV use of Orencia in children < 6Y has not been established. The safety and efficacy of Orencia pre-filled pen for SC use in children < 18Y has not been established. The safety and efficacy of Orencia pre-filled syringe in children < 2Y has not been established.
CONTRAINDICATIONS: Hypersensitivity to the active substance or excipients. Severe and uncontrolled infections such as sepsis and opportunistic infections.
WARNINGS AND PRECAUTIONS: Allergic Reactions: Caution in patients with a history of allergic reactions. Anaphylaxis or anaphylactoid reactions can occur after the first infusion and can be life threatening. Orencia I.V. or SC should be discontinued permanently if a patient develops serious allergic or anaphylactic reaction. Infections: Caution should be exercised when considering use in patients with a history of frequent infections, or underlying conditions which may predispose to infection. Treatment with Orencia should not be initiated with patients with active infections until infections are controlled. Anti-rheumatic therapies have been associated with hepatitis B reactivation. Screening for tuberculosis and hepatitis B should be performed prior to therapy. Any patient who develops a new infection should be closely monitored and Orencia should be discontinued if a patient develops a serious infection. Monitor patients for signs of infection when transitioning from TNF-antagonist to Orencia. Co administration of Orencia with biologic immunosuppressive or immunomodulatory agents could potentiate the effects of abatacept on the immune system. Treatment with immunosuppressive therapy may be associated with progressive multifocal leukoencephalopathy (PML). Orencia treatment should be discontinued if neurological symptoms suggestive of PML occur, and appropriate diagnostic measures initiated. Malignancies: The potential role of Orencia in the development of malignancies is unknown. However periodic skin examination is recommended for all patients, particularly those with risk factors for skin cancer. Elderly: Caution should be used when treating elderly patients due to a higher incidence of infections and malignancies in this patient group. Autoimmune processes: Theoretical risk of deterioration in autoimmune disease. Immunisation: Live vaccines should not be given simultaneously or within 3 months of discontinuation of Orencia. See SmPC.
PREGNANCY AND LACTATION: Abatacept may cross the placenta into the serum of infants born to women treated with abatacept during pregnancy. Consequently, these infants may be at increased risk of infection. Administration of live vaccines to infants exposed to abatacept in utero is not recommended for 14 weeks following the mother’s last exposure to abatacept during pregnancy. Do not use in pregnancy unless the clinical condition of the woman requires treatment with abatacept. Women of childbearing potential have to use effective contraception during treatment with Orencia and up to 14 weeks after the last dose of abatacept. Breast-feeding should be discontinued during treatment with Orencia and for up to 14 weeks after the last dose of abatacept treatment.
UNDESIRABLE EFFECTS: Refer to SmPC for full list. In clinical trials and post-marketing experience, the following adverse drug reactions were reported. Very Common (≥ 1/10): upper respiratory tract infection including tracheitis*, nasopharyngitis, sinusitis. Common (≥ 1/100 to < 1/10): lower respiratory tract infection (including bronchitis)*, urinary tract infection, herpes infections (including herpes simplex, oral herpes and herpes zoster), pneumonia*,a, influenza*, headache, dizziness, hypertension, blood pressure increased, cough, abdominal pain, diarrhoea, nausea, dyspepsia, mouth ulceration, aphthous stomatitis, vomiting, liver function test abnormal (including transaminases increased), rash (including dermatitis), fatigue, asthenia, SC only: local injection site reactions, systemic injection reactions (e.g.pruritus, throat tightness*, dyspnea*). Uncommon (≥ 1/1,000 to < 1/100): sepsis*,a, musculoskeletal infections*, basal cell carcinoma*, thrombocytopenia*, leukopenia*, hypersensitivity*, tachycardia*, bradycardia*, chronic obstructive pulmonary disease exacerbated*, bronchospasm*. Rare (≥ 1/10,000 to < 1/1,000): tuberculosis*, bacteraemia*, lymphoma*, lung neoplasm malignant*, squamous cell carcinoma*.
*Denotes serious adverse reaction
*Denotes fatal cases
For the pJIA paediatric population, a similar adverse event profile has been observed as in the RA population, with the following exceptions: Common adverse reactions: pyrexia
Two serious infections, varicella and sepsis were reported during the initial 4 month open label phase of treatment with abatacept in pJIA. During the pJIA SC study 20 month extension period, one serious adverse reaction of infection (limb abscess) was reported in the 2 to 5 year age cohort.
LEGAL CATEGORY: POM
MARKETING AUTHORISATION NUMBER AND BASIC NHS PRICE [UK only]:
Orencia: 250 mg concentrate for solution for infusion (EU/1/07/389/001) - 1 vial pack: £302.40; 50 mg - 4 pre-filled syringes with needle (EU/1/07/389/013): £483.84; 87.5 mg - 4 pre-filled syringes with needle (EU/1/07/389/014): £846.72; 125 mg - 4 pre-filled syringes with needle (EU/1/07/389/008): £1209.60; 125 mg ClickJect pre-filled pen (EU/1/07/389/011) - 4 pre-filled pens: £1209.60
MARKETING AUTHORISATION HOLDER:
Bristol-Myers Squibb Pharma EEIG
LOCAL REPRESENTATIVE IN UK: Bristol-Myers Squibb Pharmaceuticals Ltd, Uxbridge Business Park, Sanderson Road, Uxbridge, Middlesex UB8 1DH, UK. Tel: 01895 523000
LOCAL REPRESENTATIVE IN IRELAND: Bristol-Myers Squibb Pharmaceuticals uc, Plaza 254, Blanchardstown Corporate Park 2, Dublin, D15 T867, Ireland. Tel: 01 483 3625
DATE OF LAST REVISION: December 2019
ADDITIONAL INFORMATION AVAILABLE ON REQUEST 427UK1901396-01
Adverse events should be reported. Reporting forms and information can be found at: UK - www.mhra.gov.uk/yellowcard or search for MHRA Yellow Card in the Google Play or Apple App Store; Ireland - via HPRA Pharmacovigilance at www.hpra.ie; Adverse events should also be reported to Bristol-Myers Squibb via medical.information@bms.com or 0800 731 1736 (UK); 1 800 749 749 (Ireland).
Job code 427-GB-2100075. Date July 2021.