Interstitial lung disease (ILD)
Considering interstitial lung disease as an important extra articular manifestation of rheumatoid arthritis
What is ILD?
Rheumatoid arthritis (RA) is associated with a number of different pulmonary manifestations which are a major contributory factor to morbidity and mortality.1 Interstitial lung disease (ILD) is an extra articular manifestation of RA.2 3
The term ILD encompasses a wide range of conditions which cause progressive fibrosis of the lung parenchyma.2 Of the various ILD subtypes, those most commonly associated with RA are usual interstitial pneumonia and nonspecific interstitial pneumonia.2
Why is it important?
RA-ILD is one of the most serious extra-articular manifestations of RA, contributing significantly to decreased quality of life, progressive chronic disability, high utilization of healthcare resources and poorer mortality.2 4
Both rheumatoid factor (RF) and antibodies to citrullinated protein antigens (ACPA) have been linked to the development of ILD, especially when present in high titres.1
Irrespective of the presence of ILD, ACPA seropositivity predicts highly aggressive, erosive RA leading to significant, accelerated bone erosion.5 6 7 8 9 10

Co-morbid ILD is also associated with more severe RA leading to especially poor outcomes for patients with RA-ILD.2 3
Furthermore, patients with RA-ILD have a more than threefold increased risk of premature death compared to RA patients without ILD, and have a mean survival of under 3 years following diagnosis.2 11 The increased mortality can be attributed mainly to respiratory failure caused by ILD progression and infective complications.2
Implications on RA
For patients with RA who have poor prognosis, being initiated and remaining on treatment is critical to prevent disease progression and development of extra-articular complications.7 9 10 12 However, the increased risk of serious infection associated with rheumatoid arthritis is well documented with some studies suggesting a greater than 2-fold increased risk of serious infection.13 Co-morbidities affecting the respiratory tract, such as interstitial lung disease (ILD), further increase the risk of infection for patients with rheumatoid arthritis.13 14 15
Rheumatologists often need to make important treatment decisions and plan future care in RA patients with respiratory comorbidities, despite the absence of clear evidence or consensus.3
The consideration of co-morbidities is important to consultant rheumatologists. Who stated:
"Consideration of co-morbidities is an essential part of our treatment paradigm. If a patient has interstitial lung disease secondary to Rheumatoid arthritis this would narrow down our choice of therapy to either abatacept or rituximab." 16 - Consultant Rheumatologist
What do the guidelines say?
COVID-19 has highlighted the importance of considering infection risk when making treatment decisions with emergency NICE guidance suggesting that patients with RA or ILD should avoid attending face to face consultations wherever possible.17 18 The guidance goes one step further in recommending that patients receiving intravenous biologic therapy be switched to the same treatment in subcutaneous form, and where this is not possible, discussing an alternative subcutaneous treatment.17
Various studies have demonstrated that biologic molecules have different profiles of infection risk.19 20 21 The British Society for Rheumatology (BSR) biologic DMARD safety guidelines in inflammatory arthritis take account of this by recommending that use of abatacept or etanercept should be considered in patients who are at high risk of infection.16
In addition to increasing the risk of infection, evidence suggests that some biologics are also responsible for worsening RA-related pre-existing ILD.22 The BSR Guidelines also provide recommendations on the use of biologics in RA-ILD. They state that biologics should be used with caution, but rituximab or abatacept may be considered as first line therapy.16 Furthermore, patients receiving biologics should be regularly reviewed by a respiratory physician experienced in ILD.16
Implications on clinical practice
Keeping patients out of hospital is a key treatment consideration to minimise infection risk. The importance of this has somewhat been compounded by the Covid-19 pandemic.
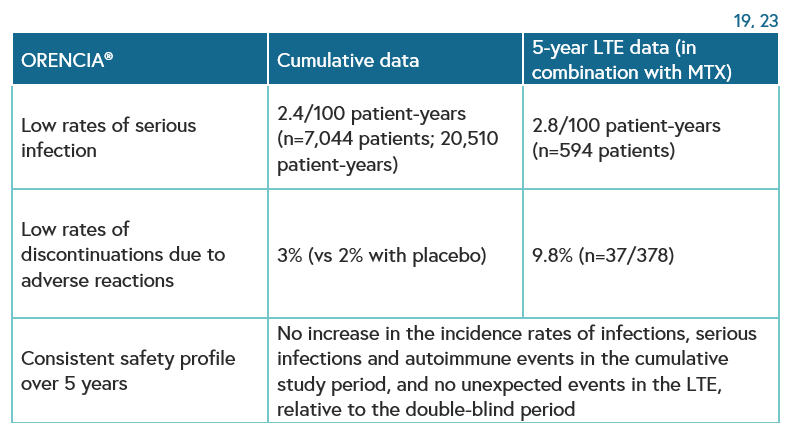
Orencia (abatacept) may be a suitable choice for patients with RA-ILD due to its acceptable long-term safety profile across clinical studies.16 Low rates of serious infections and discontinuations due to adverse reactions have been observed in patients treated with Orencia.19 23 The use of Orencia is supported by consultant rheumatologists who state:
"My treatment of choice for an RA patient with ILD is now abatacept, and I'm pleased we have BSR endorsement to use it." 16 - Consultant Rheumatologist
In MTX-refractory patients with RA, longterm abatacept treatment was well tolerated and provided a consistent safety profile and sustained efficacy, with high patient retention. Radiographic progression continued to be inhibited with ongoing treatment.23 Abatacept has been studied in patients with active rheumatoid arthritis in placebo-controlled clinical trials (2,653 patients with abatacept, 1,485 with placebo).19
Serious infections, including sepsis and pneumonia, have been reported with abatacept. Some of these infections have been fatal. Treatment with ORENCIA should not be initiated in patients with active infections until infections are controlled. Physicians should exercise caution when considering the use of ORENCIA in patients with a history of recurrent infections or underlying conditions which may predispose them to infections. Administration of ORENCIA should be discontinued if a patient develops a serious infection.
Furthermore, Orencia is supported by a Homecare programme enabling use in community settings.


